Madison and Tom spent a few days in sunny New Orleans at the ACS conference.
Madison presented a poster, connections were made and old friends were reacquainted…
We are very excited to open the application process for Summer 2024!
Lots of info here:
Please read this post carefully and follow directions to apply.
Go to careers.msu.edu. Search for job # 911371. Follow instructions to apply.
Please include with your application:
Please email Laura Ives at iveslaur@msu.edu with any questions. <—— that is me. I am very responsive.
Our application window will close February 29, 2024.
We are looking forward to hearing from you!
If you have been paying attention you have seen Dennis’ name and picture pop-up on our research posts this past summer. Dennis joined us this spring and has been a valuable addition to our research team. He was a terrific mentor to our summer students and has enthusiastically embraced the lab’s research. We are lucky to have him!
Dennis has an extensive resume, here are a few highlights:
Thank goodness he is here now working with us. His experience and insights are valuable and his guidance for our future researchers is second-to-none. We appreciate you Dennis!
Optimizing the molecular design of pyridinium anolytes for energy storage applications
Madison Shaffer and Dr. Thomas Guarr
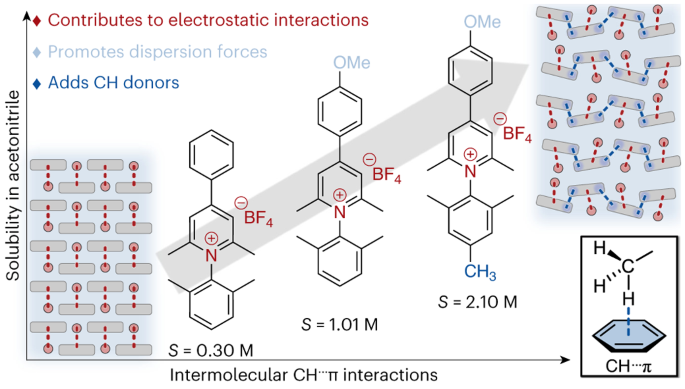
Redox flow batteries (RFBs) offer a safe and environmentally friendly alternative for grid-scale energy storage applications. The organic materials used in these batteries must meet certain criteria in order for the battery to work efficiently. Specifically, the anolyte must have a low reduction potential, stable reduced state, and high solubility. A well-known pyridinium salt, methyl viologen, is commonly used as an anolyte, and previous work in our lab has led to the discovery of new pyridinium salts that offer lower reduction potentials and greater stability. However, the solubility of these systems is typically low, limiting the energy density of RFBs incorporating such materials. It has recently been discovered that small changes in molecular structure can cause an increase in C-H – pi-interactions, which results in a dramatic increase in solubility (1). This work emphasizes the optimization of the molecular design of pyridinum anolytes and the effects it has on overall solubility.
Madison graduated from Alma College (another one!) this spring with a degree in Chemistry. We are really excited that she has decided to stick around our lab for a while and continue to help us with some great research. Thanks Madison! I hope you enjoy it here. We love having you.
Evaluating ionic liquid pyridinium compounds for non-aqueous redox flow batteries
Ed Zornow, Dennis Downing and Dr. Thomas Guarr
The energy capabilities of aqueous Redox Flow Batteries (RFBs) are limited by the use of water as a solvent which has a narrow potential window, resulting in relatively low energy density. In addition, many redox-active compounds of interest are not highly soluble in aqueous electrolytes. Using non-aqueous electrolytes in RFBs can circumvent these issues. Ionic liquids are organic salts that are in the liquid state at room temperature and exhibit attractive electrolyte properties such as wide potential windows, and high conductivity. Furthermore, by using a redox-active compound as the basis, solubility is maximized since there is no additional solvent or electrolyte salt. Functionalizing pyridinium compounds with hydrocarbon or oligoether chains to form ionic liquid derivatives has the potential to provide effective and recyclable electron carriers for non-aqueous redox-flow batteries. Several pyridinium compounds of various functionalizing groups with hydrocarbons were synthesized and characterized via NMR, mass spectrometry, and cyclic voltammetry.
Ed graduated in the spring with a Chemistry degree from Notre Dame. He spent the summer synthesizing pyridinium compounds in our lab. Ed is a great guy and we are waiting to hear what his future holds.
Synthesis of novel carbazole derivatives for electrochemical applications
Daniel Swanson, Dennis Downing, and Thomas Guarr
Carbazoles are of particular interest for electrochemical applications such as redox-flow batteries (RFBs) because of their ability to undergo reversible electrochemical oxidation at high potentials. Polymerization of the carbazole radical cation decreases reversibility but can be subdued by blocking areas of high electron-spin density (1-,3-,6-, and 8-positions). Functionalization with electron-withdrawing groups has been shown to cause an increase in oxidation potential. Moreover, the addition of alkyl groups distorts the preferred planar geometry of the radical cation. Using an intramolecular oxidative coupling route previously discovered in our lab, we synthesized several carbazole derivatives bearing electron withdrawing groups at the 3- and 6-positions and methyl groups at the 1- and 8-positions. These carbazole derivatives were synthesized with the eventual goal of creating linked anolyte-catholyte materials for RFBs.
Daniel graduated with a Chemistry degree from Central Michigan University this spring, and joined us for the summer before he started at Michigan State University in East Lansing to study a Masters in Chemical Engineering. Daniel has a very bright future and he will be great at anything he wants to do. We appreciate that you spent the summer with us Daniel!
Novel Dendrimer synthesis for use in non-aqueous redox flow batteries
Alexandra Glennon, Dennis Downing and Dr. Thomas Guarr
This research is focused on nonaqueous flow batteries, specifically the membrane barrier which acts as a wall keeping the electrolyte solutions separate. Nonaqueous batteries can contain a membrane which is less expensive than those in aqueous batteries, but it is not as ‘flow-proof’, meaning that molecules can still travel through it if small enough. A possible solution to this problem is dendrimers. Dendrimers are tree-like molecules with multiple ‘branches’ that can be attached to molecules to make them larger while also increasing electron density. This larger molecule then inhibits the electrolytes’ ability to move through the membrane barrier separating the electrolytic solutions, allowing only the electrons in the solution to cross over and creating a flowing electrochemical battery. the dendrimer used in this research was attached to pyryliums in order to increase molecular size and then analyzed through mass spectrometry, proton NMR, TLC, cyclic voltammetry and differential pulse voltammetry. Currently, pyridinium compounds are being synthesized in hopes to make additional dendrimers with them.
Alex completed the third year of her Chemistry degree at the University of Iowa before she came to try out research with us this summer. She enjoyed Holland and is interested in future battery research at U of Iowa. She is completing her final year and we are excited to find out what she does when she graduates next year.
Ferrocene- based donor-acceptor systems
Taylor D. Opolka, Dennis Downing and Dr. Thomas Guarr
Redox Flow Batteries (RFBs) are preferred over traditional lithium-ion cells for grid storage applications based on increased cycle longevity and safety. However, the cell voltage of RFBs is limited by the low potential window of aqueous solvents and a relatively small library of active materials. Typical RFB design also requires the use of ion-selective membranes to prevent the mixing of anolyte and catholyte, which in turn increases cost. Employing non-aqueous solvents allows much higher cell voltages, but ion-selective membranes are highly resistive when employed in aqueous solvents. Previous work in our lab has focused on the use of bifunctional organic molecules composed of covalently lined anolytes/catholytes that possess three stable oxidation states and can use a simple, inexpensive porous separator rather than an ion-selective membrane. This work focuses on slight modifications of the aryl-linked ferrocene donor-pyridinium acceptor systems in order to improve solubility in inducing molecular asymmetry.
Taylor returned to the lab this summer after finishing up at Davenport and earning her degree and we were really happy to have her back. She is starting a PhD program at Notre Dame this fall that we have no doubt she is going to do great at. Thanks for spending the summer with us again Taylor!
Read up on our continued efforts to develop next-generation batteries to complement renewable power generation…
We are excited to announce that Tom Guarr and Charley Hengesbach from the OESLab have been published in Nature Chemistry! This is part of a collaboration with David Hickey from MSU and Jolt Energy Storage. Better solubility of our electrochemical compounds = better batteries!
Grid-scale energy storage applications, such as redox flow batteries, rely on the solubility of redox-active organic molecules. Although redox-active pyridiniums exhibit exceptional persistence in multiple redox states at low potentials (desirable properties for energy storage applications), their solubility in non-aqueous media remains low, and few practical molecular design strategies exist to improve solubility. Here we convey the extent to which discrete, attractive interactions between C–H groups and π electrons of an aromatic ring (C–H···π interactions) can describe the solubility of N-substituted pyridinium salts in a non-aqueous solvent. We find a direct correlation between the number of C–H···π interactions for each pyridinium salt and its solubility in acetonitrile. The correlation presented in this work highlights a consequence of disrupting strong electrostatic interactions with weak dispersion interactions, showing how minimal structural change can dramatically impact pyridinium solubility.
Congrats to Dr. Guarr, Dr. Hickey, Sharmila Samaroo and Charley Hengesbach!